Diamonds are composed of carbon atoms, held tightly together in a crystal lattice by strong covalent bonds. When they are composed of pure carbon and are without inclusions or structural defects, they are colorless.
Nitrogen atoms are very small and have the ability to substitute for the carbon atoms in diamond’s crystal structure. Trace amounts of nitrogen substituting for carbon in the diamond crystal lattice will cause the diamond crystal to selectively absorb blue light and selectively transmit yellow. This will cause those nitrogen-bearing diamonds to have a yellow color. Nitrogen is the most common impurity that substitutes for carbon and can comprise up to 1% of a diamond on the basis of mass.
Nitrogen can exist in the diamond crystal lattice in a number of ways. One way that influences color is when a single nitrogen atom is shared by four carbon tetrahedrons. This defect is known as the “C center” and is shown in the accompanying illustration. In this configuration, just one nitrogen atom per 100,000 carbon atoms can produce a noticeable yellow color in the crystal.
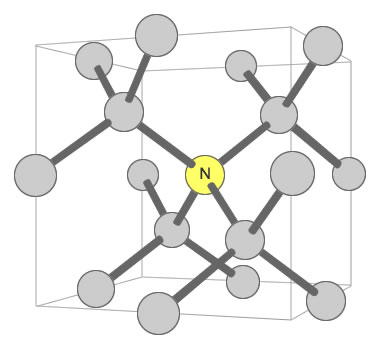
Nitrogen Substitution in Diamond: This diagram illustrates a single nitrogen atom substituting for a carbon atom in the crystal structure of a diamond. This type of substitution is known as a “C center”. This type of defect in diamond can produce a yellow color in diamond with as few as 1 nitrogen atom for every 100,000 carbon atoms.
This type of nitrogen substitution produces yellow color in lab-grown diamonds but not in mined diamonds. During the long history of natural diamonds, nitrogen atoms usually aggregate into clusters of two or more nitrogens, sometimes combined with a vacant lattice position.
When nitrogen atoms substitute for carbon in the configuration described above, it produces a defect in the diamond crystal which alters how light passes through. The defect causes a selective absorption of blue light. The remainder of the spectrum is transmitted, and that results in the perception of yellow color in the eye of the observer.
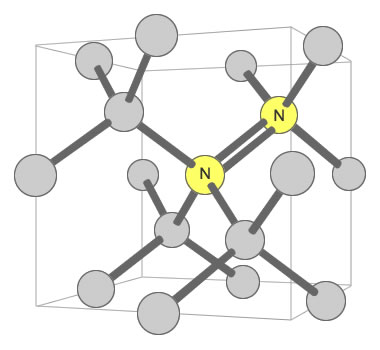
Nitrogen Pair Substitution in Diamond: This diagram illustrates two nitrogen atoms substituting for two carbon atoms in the crystal structure of a diamond. This type of substitution is known as an A-nitrogen center. This type of defect in diamond has only a weak impact on the color of the diamond.
Some types of nitrogen substitution in diamond do not produce yellow color. An example is when a pair of nitrogens substitute for carbons in two adjacent carbon tetrahedrons, with one of them shared by four tetrahedrons. This configuration is shown in a second accompanying illustration. Here, the nitrogen has almost no impact upon the color of the diamond.
A nitrogen defect that produces yellow color in many mined diamonds is the N3 defect. It consists of three nitrogen atoms clustered around a vacant carbon position in the diamond crystal lattice. When an N3 defect is accompanied by an N2 defect, certain wavelengths of blue and violet light are selectively absorbed by the diamond, and yellow light is selectively transmitted. This gives the diamond an apparent yellow color in the eye of the observer.

itrogen Pair Substitution in Diamond: This diagram illustrates two nitrogen atoms substituting for two carbon atoms in the crystal structure of a diamond. This type of substitution is known as an A-nitrogen center. This type of defect in diamond has only a weak impact on the color of the diamond.